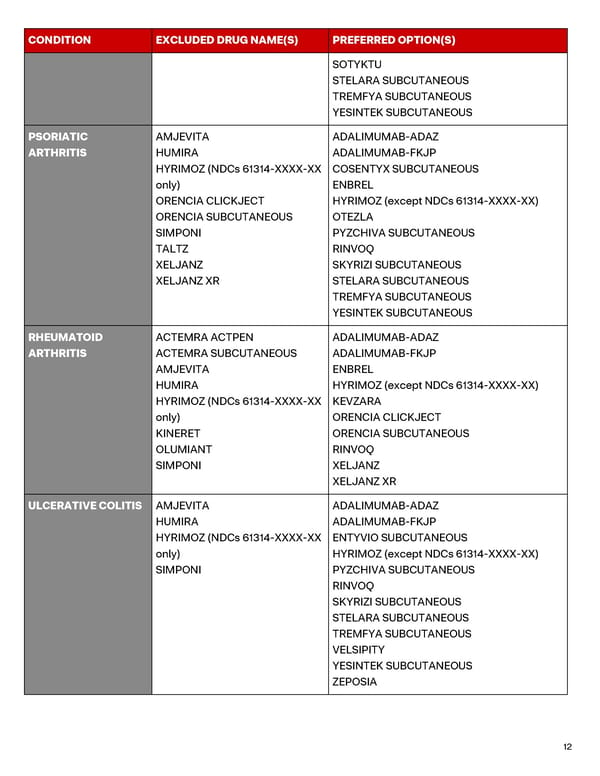
12 CONDITION EXCLUDED DRUG NAME(S) PREFERRED OPTION(S) SOTYKTU STELARA SUBCUTANEOUS TREMFYA SUBCUTANEOUS YESINTEK SUBCUTANEOUS PSORIATIC ARTHRITIS AMJEVITA HUMIRA HYRIMOZ (NDCs 61314-XXXX-XX only) ORENCIA CLICKJECT ORENCIA SUBCUTANEOUS SIMPONI TALTZ XELJANZ XELJANZ XR ADALIMUMAB-ADAZ ADALIMUMAB-FKJP COSENTYX SUBCUTANEOUS ENBREL HYRIMOZ (except NDCs 61314-XXXX-XX) OTEZLA PYZCHIVA SUBCUTANEOUS RINVOQ SKYRIZI SUBCUTANEOUS STELARA SUBCUTANEOUS TREMFYA SUBCUTANEOUS YESINTEK SUBCUTANEOUS RHEUMATOID ARTHRITIS ACTEMRA ACTPEN ACTEMRA SUBCUTANEOUS AMJEVITA HUMIRA HYRIMOZ (NDCs 61314-XXXX-XX only) KINERET OLUMIANT SIMPONI ADALIMUMAB-ADAZ ADALIMUMAB-FKJP ENBREL HYRIMOZ (except NDCs 61314-XXXX-XX) KEVZARA ORENCIA CLICKJECT ORENCIA SUBCUTANEOUS RINVOQ XELJANZ XELJANZ XR ULCERATIVE COLITIS AMJEVITA HUMIRA HYRIMOZ (NDCs 61314-XXXX-XX only) SIMPONI ADALIMUMAB-ADAZ ADALIMUMAB-FKJP ENTYVIO SUBCUTANEOUS HYRIMOZ (except NDCs 61314-XXXX-XX) PYZCHIVA SUBCUTANEOUS RINVOQ SKYRIZI SUBCUTANEOUS STELARA SUBCUTANEOUS TREMFYA SUBCUTANEOUS VELSIPITY YESINTEK SUBCUTANEOUS ZEPOSIA
 CVS Caremark Advanced Control Specialty Formulary Guide Page 11 Page 13
CVS Caremark Advanced Control Specialty Formulary Guide Page 11 Page 13